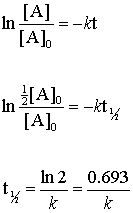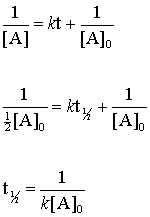half life formula for zero order reaction
Note that this equation has the form. This class of study uses to derive the half-life equation formulas in chemical kinetics reaction.

Zero Order Reactions Video Kinetics Khan Academy
A zeroth-order reaction that takes place in the.
. A 12 A₀or A₀2. It is essential to note that the half-life formula of a reaction varies with the reactions order. For the first-order reaction the half-life is defined as t 1.
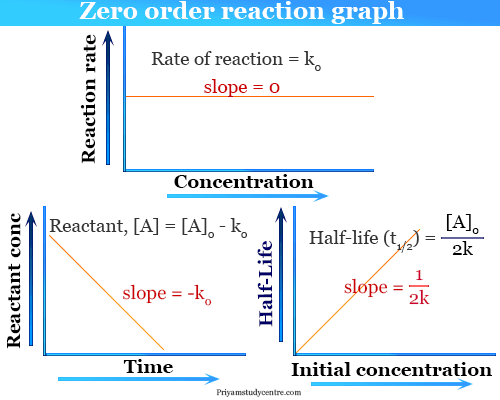
Rate k C12H22O11 Half-Life of a reaction t12. The rate constant for the reaction can be determined from the slope of the line which is equal to -k. The mathematical expression that can be employed to determine the half-life for a zero-order reaction is t 12 R 0 2k.
Half life is a particular phenomenon that takes place every day in various chemical reactions as well as nuclear reactions. The integrated rate law for the zero-order reaction A products is A_t -kt A_0. From the above-integrated equation we have.
Therefore a plot of A versus t will always yield a straight line with a slope of. It is clearly visible from the above equation that the half-life of the reaction is dependent on the rate constant as well as the initial concentration of the reactant. K t 12 12 A 0.
The Half-Life of Zero Order Reaction calculator computes the half-life in nuclear decay for a zero order reaction. As for all reaction orders the half-life for a zero-order reaction is inversely proportional to its rate constant. Hence The half-life of a reaction t½ occurs when consider A as a reactant molecule.
Therefore tt ½ x x2. It is the time in which the concentration of a reactant is reduced to one half of its initial concentration. 1A n-1 1 A 0 n-1 n-1 kt.
Half life in zero order reaction. K-k k. Where A 0 Initial concentration of reactant at timet 0.
T 12 0693k. The order of the reaction or enough information to determine it. Half life means 50 percent of reactants disappear in that time interval.
In some cases we need to know the initial concentration A o Substitute this information into the equation for the half life of a reaction with this order and solve for t ½. We know that at the half-life time eqt_12 eq the concentration of the reactant will. For a general reaction.
Half life formula for nth order reaction. If we set the time t equal to the half-life the corresponding concentration of A at this time is equal to one-half of its initial concentration. Remember that the formula for the half-life of a reaction varies along with changing the order of the reaction.
The rate constant k for the reaction or enough information to determine it. Half-life of a first. Now replacing t with half-life t12 in the above equation.
As for other reaction orders an equation for zero-order half-life may be derived from the integrated rate law. The rate constant for a zero-order reaction is 054 M-1s-1. A reactions half-life formula changes depending on the order of the reactions.
Read More Second order Reaction. Half-life refers to the amount of time it takes for half of a particular sample to react. 12 A A 0 - k t 12.
We can derive an equation for determining the half-life of a first-order reaction from the alternate form of the integrated rate law as follows. As for other reaction orders an equation for zero-order. In each succeeding half-life half of the remaining concentration of the reactant is consumed.
Thus for a first-order reaction each successive half-life is the same length of time as shown in. And half-life is usually measured in seconds. NA Product The rate law of zero order kinetics is.
Y m x ymx y m x. T ½ x2k where x initial concentration of reactant. The half-life of a reaction t12 is the time required for one-half of a given amount of reactant to be consumed.
The half-life of a zero-order reaction the formula is given as t12 R02k The half-life of a first-order reaction is given as t12 0693k The half-life of a second-order reaction is given by the formula 1kR0. T 12 is the half-life of. The half-life formula for various reactions is given below.
Half-life is denoted by t½. R k r kNH 3 0. Half-Life of a Zero Order Reaction.
Substituting these terms into. It is to be noted that the formula for the half-life of a reaction varies with the order of the reaction. Half-Life of a Zero-Order Reaction The half-life of a reaction describes the time needed for half of the reactants to be.
This is the integrated rate law for a zero-order reaction. Thus for a first-order reaction each successive half-life is the same length of time as shown in. Using the concentration-time equation for a second-order reaction we can solve for half-life.
T 12 A 0 2k. A zero order reaction implies that the rate of the reaction does not depend on the concentration of the reactant. A reaction is zero order if its rate is independent of the concentration of reactants or a zero order reaction means that the rate of reaction is proportional to zero.
For the 1 st order reaction the half-life is. The rate constant for a zero-order reaction is. Because this equation has the form y mx b a plot of the concentration of A as a function of time yields a straight line.
The integrated rate law in the zero-order kinetics uses to derive half-life equations in chemistry x k 0 t. The half-life of a Zero-th order reaction is t A0 2kHere I. Half Life Method other methods JEENEET - Duration.
T 12 12 k A 0. T ½ 0693 k For a second order reaction 2A products or A B products when A B rate kA 2. It is represented by t12.
The Half-Life of Zero Order Reaction calculator computes the half-life in nuclear decay for a zero order reaction. A A 0 - kt. Learn the half life formula here.
For a zero-order reaction the integrated rate law is. Using the decomposition of hydrogen peroxide see this lesson as an example we find that during the first half. However the half-life of a zero-order reaction increases as the initial concentration increases.
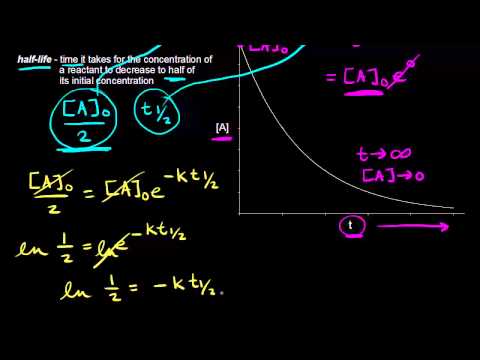
Half Life Of A First Order Reaction Video Khan Academy

Summary Of The Kinetics Of Zero Order First Order Ppt Download

Kinetics And Drug Stability Ed

Principles And Kinetics Of Drug Stability Phr 416 Ppt Video Online Download

Half Life Of Zero Th 0th Order Reaction Derivation Youtube

Half Life Expressions Chemistnate

Integrated Rate Laws Zero First Second Order Reactions Chemical Kinetics Youtube

Kinetics Order Of Reactions Ppt Video Online Download

Zero Order Reactions Chemistry Class 12 Iit Jee Main Advanced Neet Aipmt Askiitians Youtube

Half Life Of A Zero Order Reaction Is 250sec T75 T100 Of The Reaction Respectively In Sec Are Edurev Neet Question

Half Life Expressions Chemistnate